This article disappeared…our bad, below a recap.
Lead acid batteries are still the most common large batteries around. They are found in every car and have seen remarkably little innovation in the past decades. They can be used for large capacity storage, but they still wear down and there is no easy modular recycling infrastructure for worn out electrodes, most are still throwaway. We hear a lot about flow batteries for large capacity storage. Flow batteries are batteries where the electrolyte reservoiw is much bigger than usual. Most of it is out of reach of the electrodes most of the time, so a pump needs to cause a flow so it can deliver its charge. Flow batteries are resonably simple devices, we have written about the Vanadium type, a type that is unpatented and works like a charm over long periods without intervention. Vanadium has two compartments with electrolyte as shown below.
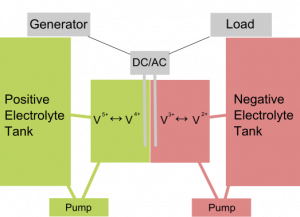
Below a summary of the work at the University of Southampton in the UK. There a lead acid flow battery was build and tested on a small scale. Scalability is one thing flow batteries are designed for though.
“Technology based on flow batteries is one potential solution to such energy storage because they have a very high energy efficiency. In Southampton, the programme centres on the soluble lead flow battery, Click on the image to see the electrode reactions.”

“And the electrolyte is methanesulfonic acid. Compared to other flow batteries, the advantage is that a separator is unnecessary and there is only one electrolyte flow. The electrode reactions are possible at high rates. The challenge is now to control the deposition of the lead and lead dioxide at all the rates and in all the conditions met in an operating energy storage system.”

(source)
As you can see above the battery performs over many cycles with little loss. The loss that occurs gradually is usually a result of degradation of the electrodes and sediment in the electrolyte. So these aspects require maintenance. Every other type of energy infrastructure requires maintenance as well though. So why isn’t this tried?